 :max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
All you really need to find is something called the mass number. The atomic weight is actually a weighted average of all of the naturally occurring isotopes of an element relative to the mass of carbon-12. The atomic weight is basically a measurement of the total number of particles in an atom's nucleus. The 35 remaining electrons were outnumbered by the 36 positively charged protons, resulting in a charge of +1. It is positively charged because a negatively charged electron was removed from the atom. The plus sign means that this is a positively charged ion. Adding or removing electrons from an atom does not change which element it is, just its net charge.įor example, removing an electron from an atom of krypton forms a krypton ion, which is usually written as Kr +. An ion is nothing more than an electrically charged atom. 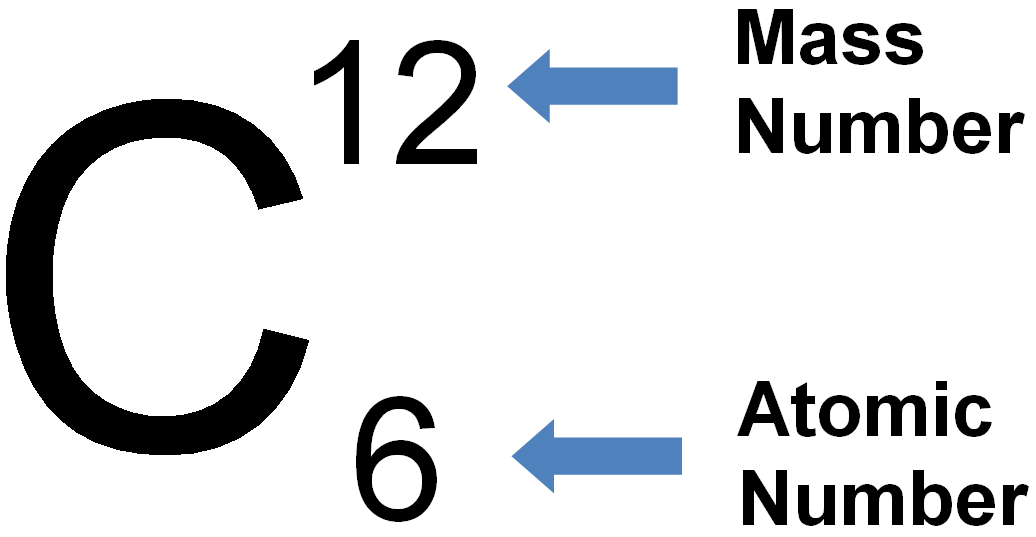
If you need to know how the electrons are arranged around an atom, take a look at the ' How do I read an electron configuration table?' page.Īn atom can gain or lose electrons, becoming what is known as an ion. In our example, an atom of krypton must contain 36 electrons since it contains 36 protons.Įlectrons are arranged around atoms in a special way. Atoms must have equal numbers of protons and electrons. That means that there must be a balance between the positively charged protons and the negatively charged electrons. For example, removing one proton from an atom of krypton creates an atom of bromine.īy definition, atoms have no overall electrical charge. Adding or removing protons from the nucleus of an atom creates a different element. If an atom doesn't have 36 protons, it can't be an atom of krypton. The interesting thing here is that every atom of krypton contains 36 protons. This tells us that an atom of krypton has 36 protons in its nucleus. In our example, krypton's atomic number is 36. The atomic number is the number of protons in an atom of an element. The atomic number is the number located in the upper left corner and the atomic weight is the number located on the bottom, as in this example for krypton: Use the Table of Elements to find your element's atomic number and atomic weight. If it makes things easier, you can select your element from an alphabetical listing. Go to the Periodic Table of Elements and click on your element. The first thing you will need to do is find some information about your element. To find the number of protons, electrons and neutrons in an atom, just follow these easy steps: This would mean that indium’s atomic mass was actually 113, placing the element between two other metals, cadmium, and tin.How many protons, electrons and neutrons are in an atom of krypton, carbon, oxygen, neon, silver, gold, etc.? Because elemental indium is a silvery-white metal, however, Mendeleev postulated that the stoichiometry of its oxide was really In 2O 3 rather than InO. 
If this atomic mass were correct, then indium would have to be placed in the middle of the nonmetals, between arsenic (atomic mass 75) and selenium (atomic mass 78). The atomic mass of indium had originally been reported as 75.6, based on an assumed stoichiometry of InO for its oxide. He discovered, for example, that the atomic masses previously reported for beryllium, indium, and uranium were incorrect. When the chemical properties of an element suggested that it might have been assigned the wrong place in earlier tables, Mendeleev carefully reexamined its atomic mass. The observed properties of gallium and germanium matched those of eka-aluminum and eka-silicon so well that once they were discovered, Mendeleev’s periodic table rapidly gained acceptance. Two of the blanks Mendeleev had left in his original table were below aluminum and silicon, awaiting the discovery of two as-yet-unknown elements, eka-aluminum and eka-silicon (from the Sanskrit eka, meaning “one,” as in “one beyond aluminum”). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
